Prove performance. Expand reach.
Showcase your packaging in real-world pharma operations — verified, comparable, and visible to every decision-maker in the industry.


Showcase your packaging in real-world pharma operations — verified, comparable, and visible to every decision-maker in the industry.









.webp?width=2251&height=920&name=Pfizer_(2021).webp)
Validaide brings your packaging solutions to the world’s largest pharma logistics networks.
Be considered in more lane risk assessments, accelerate qualification processes, and demonstrate performance more clearly.
36
Packaging manufacturers
1,345
Packaging solutions
100%
traceable documentation
Pharma manufacturers and forwarders increasingly select packaging based on proven performance, validation speed, and quality documentation. Without standardization and visibility, even strong solutions risk being overlooked.



Validaide makes your packaging solutions visible, comparable, and trusted in real pharma transport decisions.
Position your packaging where planning decisions happen. Publish verified performance data in a harmonized format used directly in LRAs, so pharma and logistics partners can evaluate, compare, and select solutions with confidence.
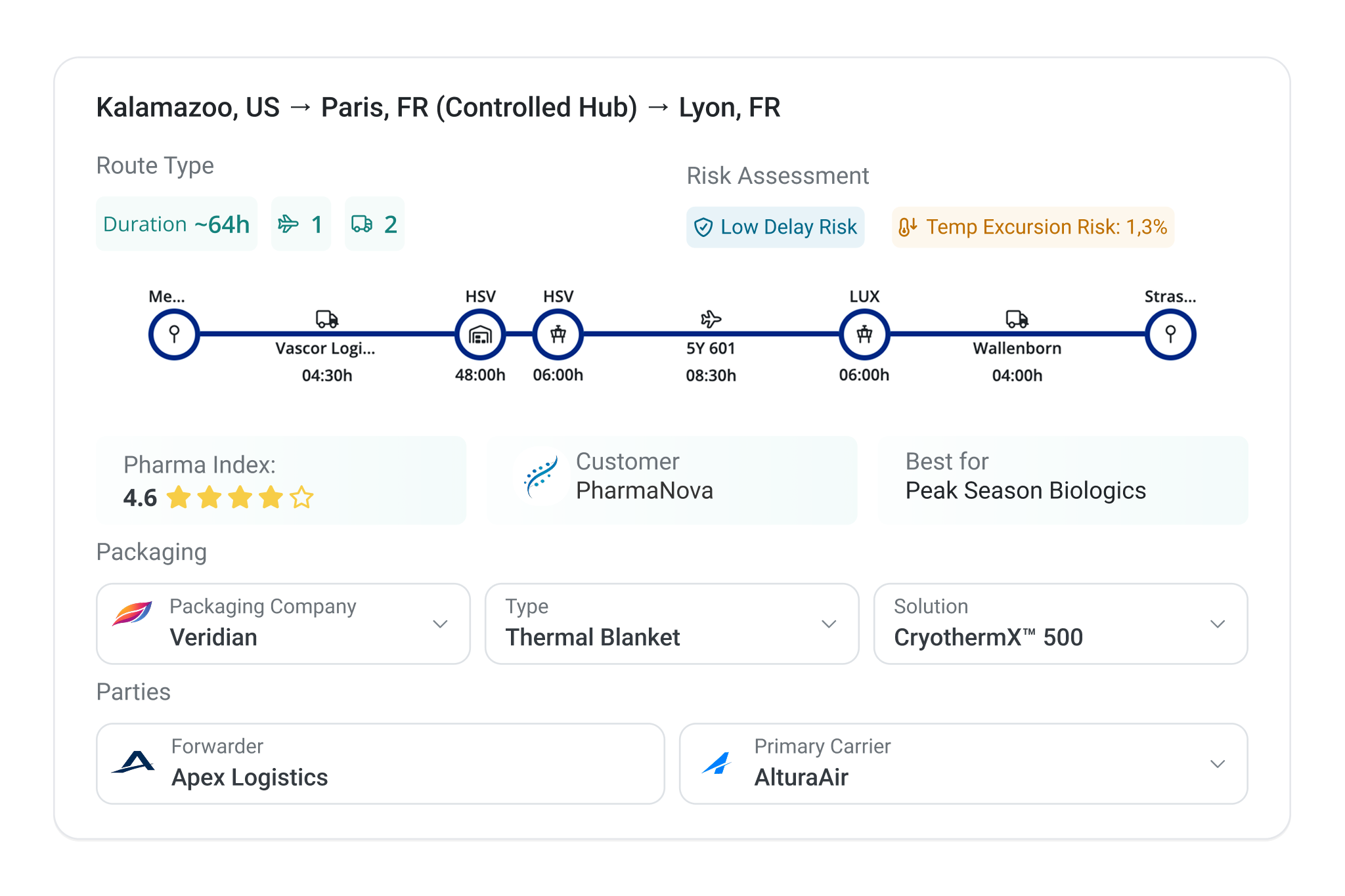
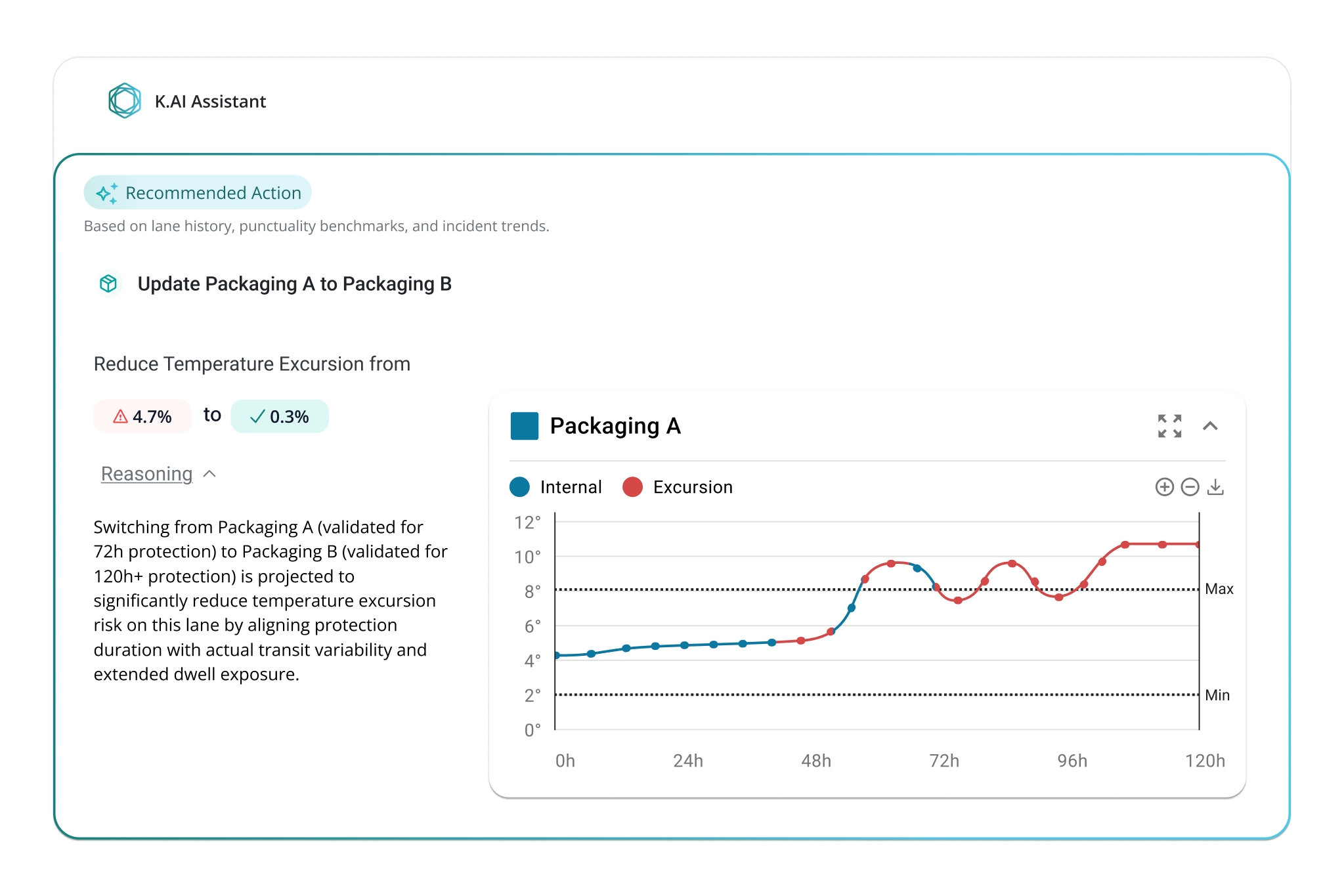
Make instructions for use, handling requirements, and performance evidence available where shipments are planned, monitored, and reviewed. This supports correct usage, faster deviation handling, and confident audits without repeated requests or manual follow-up.
Guided onboarding, role-based training, and ongoing change support help teams realize the full potential of Validaide in their daily work. Targeted projects for qualification, performance validation, and CO2 insights strengthen customer proposals and translate technical excellence into commercial advantage
